
The study of meteorology was born when these oscillations were explained as manifestations of changes in atmospheric pressure. Small variations in height that happened in barometers were familiar to scientists at the time. He is recognized as the first to explain atmospheric pressure in a modern way. When Torricelli presented the first mercury barometer to the wider population, he drew interest. It was named torr in honor of Evangelista Torricelli, an Italian mathematician and physicist who established the barometer principle in 1644. The torr (symbol: Torr) is a pressure unit defined as exactly 1/760 of a familiar atmosphere on an absolute scale (101325 Pa). Furthermore, mmHg and torr are almost the same units. What is a Torr Unit?īecause the early barometers were made of mercury (Hg) inside a vertical glass, the natural pressure unit was mmHg (or mm Hg), which stands for a millimetre of mercury. Also, try Volume Conversion tool for conversion between different volume units, or maybe convert Grams to Calories. In torr, what is the atmospheric pressure? How many torrs equal one atm? How much is 750 torr to atm, or maybe 720 torr to atm? Is there any pressure conversion table that would make our lives easier? If you’ve ever wondered about one of these topics, you’ve come to the correct place!Ĭheck our other conversion tools, such as Knots to mph, Time Unit Converter to convert between different units of time. 
The article includes a definition of the torr unit and correlations between torr and atm (unit of pressure). /rebates/&252ffrom-mmhg-to-atm. Symbols, abbreviations, or full names for units of length,Īrea, mass, pressure, and other types.This is the Torr to Atm Conversion tool for converting torrs to atms (standard atmospheres). You can find metric conversion tables for SI units, as wellĪs English units, currency, and other data. It is approximately equal to Earth's atmospheric pressure at sea level.Ĭonversion calculator for all types of measurement units. It is sometimes used as a reference pressure or standard pressure. The standard atmosphere (symbol: atm) is a unit of pressure defined as 101325 Pa (1.01325 bar). This small difference is negligible for most applications outside metrology. The difference between one millimeter of mercury and one torr, as well as between one atmosphere (101.325 kPa) and 760 mmHg (101.3250144354 kPa), is less than one part in seven million (or less than 0.000015%). You can view more details on each measurement unit: mmHg or atm The SI derived unit for pressure is the pascal. We assume you are converting between millimeter of mercury 0 C and atmosphere standard. 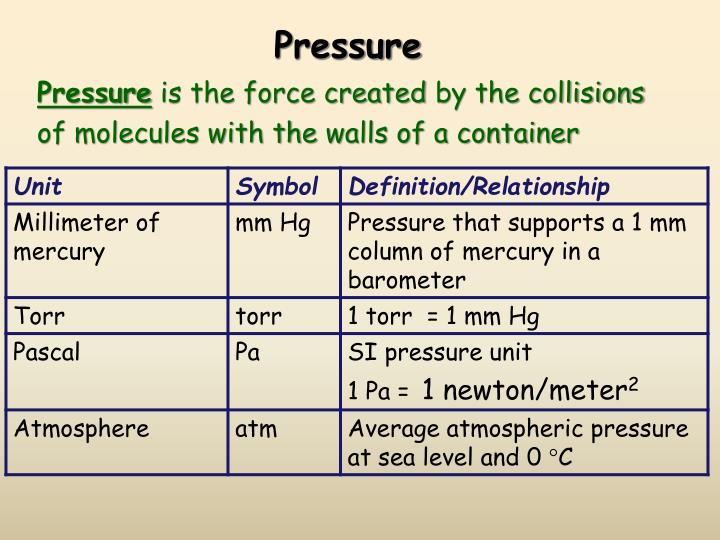
The relationship between the torr and the millimeter of mercury is: How many mmHg in 1 atm The answer is 759.9998769899. The decimal form of this fraction is approximately 133.322368421. 
Therefore, 1 Torr is equal toġ01325/760 Pa. The torr is defined as 1/760 of one standard atmosphere, while the atmosphere is defined as 101325 pascals. Examples include gas pressure conversions torr to atm. Express this pressure in atmospheres, kilopascals, torrs, pounds per square inch, and pascals. This video tutorial explains how to convert gas pressure units such as torr, atm, mm Hg, kpa, and psi. The millimeter of mercury by definition is 133.322387415 Pa (13.5951 g/cm3 × 9.80665 m/s2 × 1 mm), which is approximated with known accuracies of density of mercury and standard gravity. A trick question Example 12: A student reads a barometer in the laboratory and finds the prevailing atmospheric pressure to be 720.0 mmHg. You can do the reverse unit conversion fromĪtm to mm Hg, or enter any two units below: Enter two units to convert From:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
